Press Releases

For Spains Seniors Healthy Plant-Based Diets Cut Mortality Risks
Despite the rising global popularity of healthy pro-vegetarian diets (PVG), including the Mediterranean diet and...

Over Low-Carb Diets Study finds Low and High GI Diets Boost Endurance Performance
Study conducted a randomized controlled trial to evaluate how varying carbohydrate intake and the glycemic...

Study Finds Childhood Trauma Linked to Higher Rates of Somatic Symptoms
A group of researchers investigated the prevalence and severity of somatic symptoms among children and...

In Dementia Patients Study Shows Antipsychotic Drugs Increase Health Risks
Researchers assess the adverse effects associated with the use of antipsychotic drugs in people with...
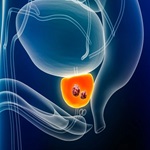
In Prostate Cancers Vulnerability to Ferroptosis New Research Pinpoints Key Pathways
Researchers examined the molecular mechanisms and metabolic processes that drive ferroptosis — a form of...

In Americans Study Reveals Plant Proteins Impact on Nutrient Levels
Researchers explored the impact of increasing dietary plant protein quartiles on nutritional adequacy in the...
On Body Fat Metabolism Gut Bacteria Play a Pivotal Role in Obesitys Impact
Researchers explored how the dysregulation of gut microbiota in obesity impacts adipose tissue (AT) metabolism...

In Fasting Glucose there is Continuous Glucose Monitoring and Intrapersonal Variability
Researchers examined fasting glucose (FG) variability in nondiabetic adults using continuous glucose monitoring (CGM), assessing...

Plant-based Substitutes Health Claims Challenged in New Study
Study analyzed the effects of animal-based meat diets (ABMD) relative to PBMA diets (PBMD) on...
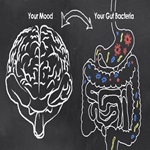
Brighten Moods and Fight Depression could be by Gut-Friendly Psychobiotics
Researchers investigated the psychobiotic treatment of depression by restoring microbial balance and regulating the microbiome-gut-brain...